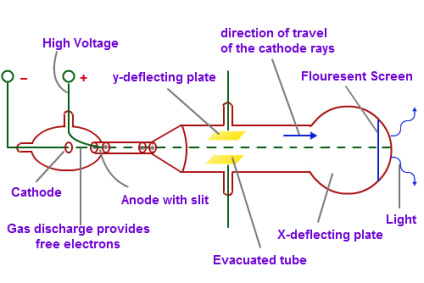
In the early 19th century physicists were trying to understand the structure of the atom and at that time J.J Thompson a British physicist conducted an experiment with the cathode ray tubes. The electron is denoted by e- and has one negative elementary charge. 
They are thought to be elementary particles of atoms as they have no known components or structure. Electron is a subatomic particle that belongs to the first generation of the lepton particle family. Every atom of any given element will have the same physical properties due to the fact that it contains exactly the same subatomic particle as that of the other one. Every atom is made up only of these three particles. But when we break the atom further it will give us electrons, protons, and neutrons. Thomson, through his now-famous cathode ray tube experiments, led to the discovery of electrons as being a part of subatomic particles, i.e., particles which make up the atom.Ītoms are the smallest particle of an element that will have the same property of the element which can never be created or destroyed according to Dalton's atomic theory. However, all that changed when in 1897, the English physicist J.J. The term ‘atom’ being wholly representative of that thought since it comes from the Greek word meaning indivisible. For a long time, the atom was considered to be the final structure of an element. Electrons are subatomic particles that, along with protons and neutrons, come to form the most fundamental part of atoms.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
